 
We are all aware that expansion of liquid water to ice exerts a tremendous force. Once the oceans filled with ice, life there would not be possible. If water did not expand when freezing, then it would be denser than liquid water when it froze therefore it would sink and fill lakes or the ocean from bottom to top. For example, fish in a pond survive the winter because ice forms on top of a pond (it floats) and effectively insulates (does not conduct heat from the pond to the atmosphere as efficiently) the rest of the pond below, preventing it from freezing from top to bottom (or bottom to top). This latter property is very important for organisms in the oceans and/or freshwater lakes. For this reason, ice floats on the water (like an ice cube in a glass of water). In fact, its volume is a little over 9% greater (or density ca. Water expands when it freezes making it less dense than the water from which it freezes. Water, however, is unique in that it bonds in such a way that the molecules are held farther apart in the solid form (ice) than in the liquid. For nearly every known chemical compound, the molecules are held closer together (bonded) in the solid state (e.g., in mineral form or ice) than in the liquid state. When water freezes, however, bonds are formed that lock the molecules in place in a regular (hexagonal) pattern. This effect also helps explain why you are supposed to add salt to ice when making ice cream or to add salt to water when cooking spaghetti (although, in this case, the effect on boiling point is minor and the added salt is mainly for flavor). Increasing salinity also lowers the temperature of maximum density. Normal seawater freezes at -2˚ C, 2˚ C colder than pure water. Incidentally, increasing salinity increases the boiling point and decreases the freezing point. As you add salts to seawater, you also change some other properties. 
The density of average seawater with a salinity of 35 o/oo (35 g/kg) and at 4˚ C is 1.028 g/cm3 as compared to 1.000g/cm3 for pure water. Table 14.1 - Densities of Some Common Substances SolidsĪs you can see by examining Table 14.However, as dissolved solids are added to pure water to increase the salinity, the density increases. Plasma will not be discussed in depth in this chapter because plasma has very different properties from the three other common phases of matter, discussed in this chapter, due to the strong electrical forces between the charges. At high temperatures, molecules may disassociate into atoms, and atoms disassociate into electrons (with negative charges) and protons (with positive charges), forming a plasma. There exists one other phase of matter, plasma, which exists at very high temperatures. In this chapter, we generally refer to both gases and liquids simply as fluids, making a distinction between them only when they behave differently. When placed in an open container, gases, unlike liquids, will escape. 
This makes gases relatively easy to compress and allows them to flow (which makes them fluids). In contrast, atoms in gases are separated by large distances, and the forces between atoms in a gas are therefore very weak, except when the atoms collide with one another. Because the atoms are closely packed, liquids, like solids, resist compression an extremely large force is necessary to change the volume of a liquid. When a liquid is placed in a container with no lid, it remains in the container. That is, liquids flow (so they are a type of fluid), with the molecules held together by mutual attraction. This occurs because the atoms or molecules in a liquid are free to slide about and change neighbors. Liquids deform easily when stressed and do not spring back to their original shape once a force is removed. A gas must be held in a closed container to prevent it from expanding freely and escaping. (c) Atoms in a gas move about freely and are separated by large distances. 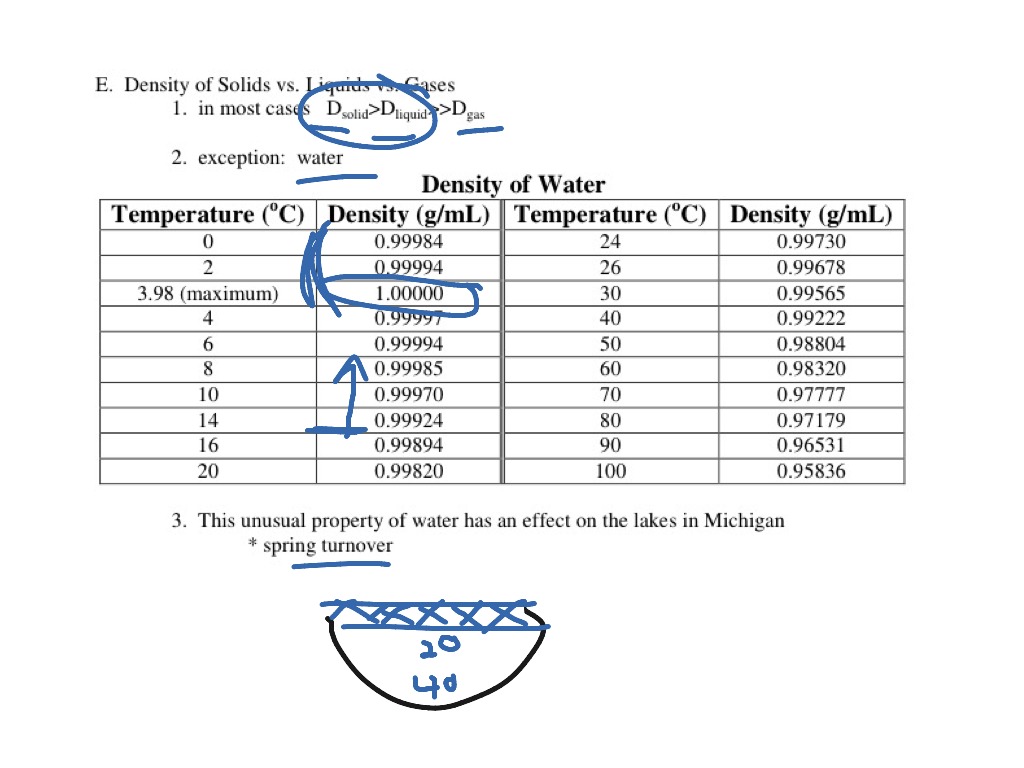
Forces between the atoms strongly resist attempts to compress the atoms. (b) Atoms in a liquid are also in close contact but can slide over one another. \): (a) Atoms in a solid are always in close contact with neighboring atoms, held in place by forces represented here by springs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
